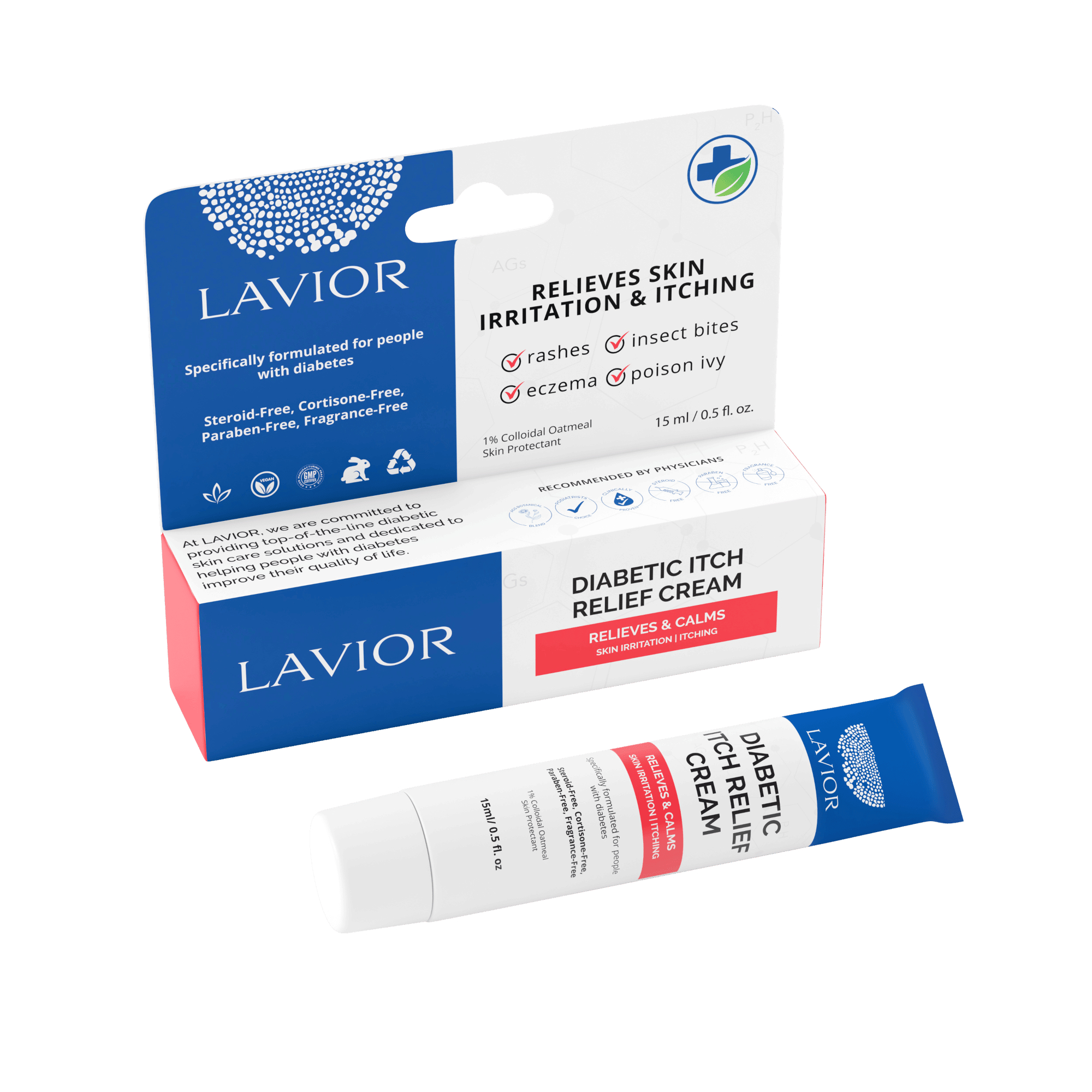
- Diabetic CareLAVIOR is a proven leader offering the most comprehensive line of diabetic wound & skincare products, from prevention to treatment.
- Wound Care and EczemaLavior offers powerful topical remedies to treat an array of wounds, burns, and chronic Eczema.
- About Us
- Media
- Contact Us
- Where to Buy
- Clinicians
Our Company
ABOUT LAVIOR
Lavior is revolutionizing skin care with its FDA-registered line of natural, physician-recommended wound and skin care products. These innovative treatments, derived from molecular isolates extracted from a novel species of medicinal plants, offer safe, effective solutions for some of the most common and challenging skin conditions.
Founded by Gilad and Anabelle Savion, Lavior was born from a vision: to harness the power of nature to heal. Their journey began with the Inula, a Mediterranean plant known as the “King of Medicinal Plants”. The Savions’ unwavering commitment to harnessing nature’s potential led to the creation of Inula AGS-RIED, a unique plant species with remarkable anti-inflammatory, anti-bacterial, antioxidant, and anti-fungal capabilities.
What sets Lavior apart?
- Breakthrough formulations: Lavior’s team of scientists has harnessed the power of botanicals and OTC active pharmaceutical ingredients to create groundbreaking formulas that outperform conventional treatments.
- Natural and gentle: Lavior products are physician-approved and hypoallergenic, making them a safe and gentle choice for even the most sensitive skin.
- Clinically proven: Backed by over 13 years of research and clinical trials, Lavior’s products deliver proven clinical safety and efficacy, consistently exceeding expectations.
- Comprehensive range: Lavior offers groundbreaking natural treatments for some of the most common and challenging skin conditions, including: diabetic ulcers / wounds and burns / eczema and atopic dermatitis / bacterial and fungal infections /skin irritations / cracked skin.

Our mission is to improve the quality of life for people with diabetes by providing innovative and natural solutions in preventive care, wound care and skin infection mitigation. This is motivated by both the current lack of effective treatments as well as emerging public concern regarding the health risks associated with products containing synthetic agents.
Our vision is to establish a new standard of care in the wound healing and diabetic skin care market. We will continue to be at the forefront of scientific research and product development so that we can provide natural, plant-based products that ease suffering, speed healing and enhance the quality of life for the hundreds of millions of consumers with diabetes.
US Certifications:
- FDA OTC – in the USA (NDC Code: 71521)
- Medical Device in the USA
- WBENC Certification as Woman-Owned Business
- Partner with ADA – Better Choices for Life
Lavior’s commitment to quality:
- cGMP-compliant manufacturing/made in the USA: Lavior products are manufactured in a state-of-the-art, cGMP FDA-compliant facility, ensuring the highest quality and safety standards.
- Stringent quality control: Lavior’s dedicated QA team closely monitors and controls every step of product development and manufacturing, guaranteeing the consistency and efficacy of their products.
Lavior is more than just a skin care company; it’s a beacon of hope for those struggling with chronic skin disorders. By harnessing the power of nature and cutting-edge science, Lavior is paving the way for a healthier, happier future for all.